Saving sight sooner makes brighter futures.
Eyerising Myopia Management Device RLRL Technology
At last, there is a simple and effective myopia control therapy developed especially for children, guardians and practitioners. Using patented technology to deliver Repeated Low-Level Red-Light (RLRL) therapy, the Eyerising Myopia Management Device unit gently stimulates blood flow in the retina to help slow the axial length’s further elongation and control myopia’s progression.
Eyerising Myopia Management Device RLRL Technology
At last, there is a simple and effective myopia control therapy developed especially for children, guardians and practitioners. Using patented technology to deliver Repeated Low-Level Red-Light (RLRL) therapy, the Eyerising Myopia Management Device unit gently stimulates blood flow in the ocular fundus to alleviate sclera hypoxia and control the axial lengthening that causes myopia.
Life changing child-friendly myopia control at home
Designed to be easy to use at home, the device is non-invasive; can be used without additional medications, eyedrops, or lenses; and is free of any known significant side effects. Children as young as 3 and up to 16 can use the device. Over 150,000 children are already using RLRL therapy each day from the comfort of home.
The child friendly go-to, in your myopia treatment toolkit
The device is a powerful addition to the clinician’s existing myopia toolkit, providing the potential for earlier treatment, additional patient treatment optionality and the potential for RLRL as a new adjunct therapy.^ The device monitors and shares patient compliance data with practictioners for use during follow-up examinations.
^ RLRL cannot be used in conjunction with atropine. Patients need to stop using atropine for 2 weeks and then start RLRL therapy.
Therapy involves looking into the device for 3 minutes of treatment, twice a day, 5 days per week, with a minimum of 4 hours between sessions. The device is controlled with a simple touchscreen and is suitable for children as young as 3 years of age.
No drugs
No injections
No contact lenses
No known significant side-effects
Streamlined reporting boosted compliance
The device is a powerful addition to the clinician’s existing myopia toolkit, removing tears and complications that limit compliance. It securely shares patient compliance data with practitioners, enabling superior follow-up assessment. It also sends reminders to the guardian when the patient misses treatment to maximise patient treatment efficacy.
Usable as a stand-alone therapy without eyedrops, lenses or complications. Or as a powerful adjunct to existing therapies for patients who are not responding to Ortho-k.
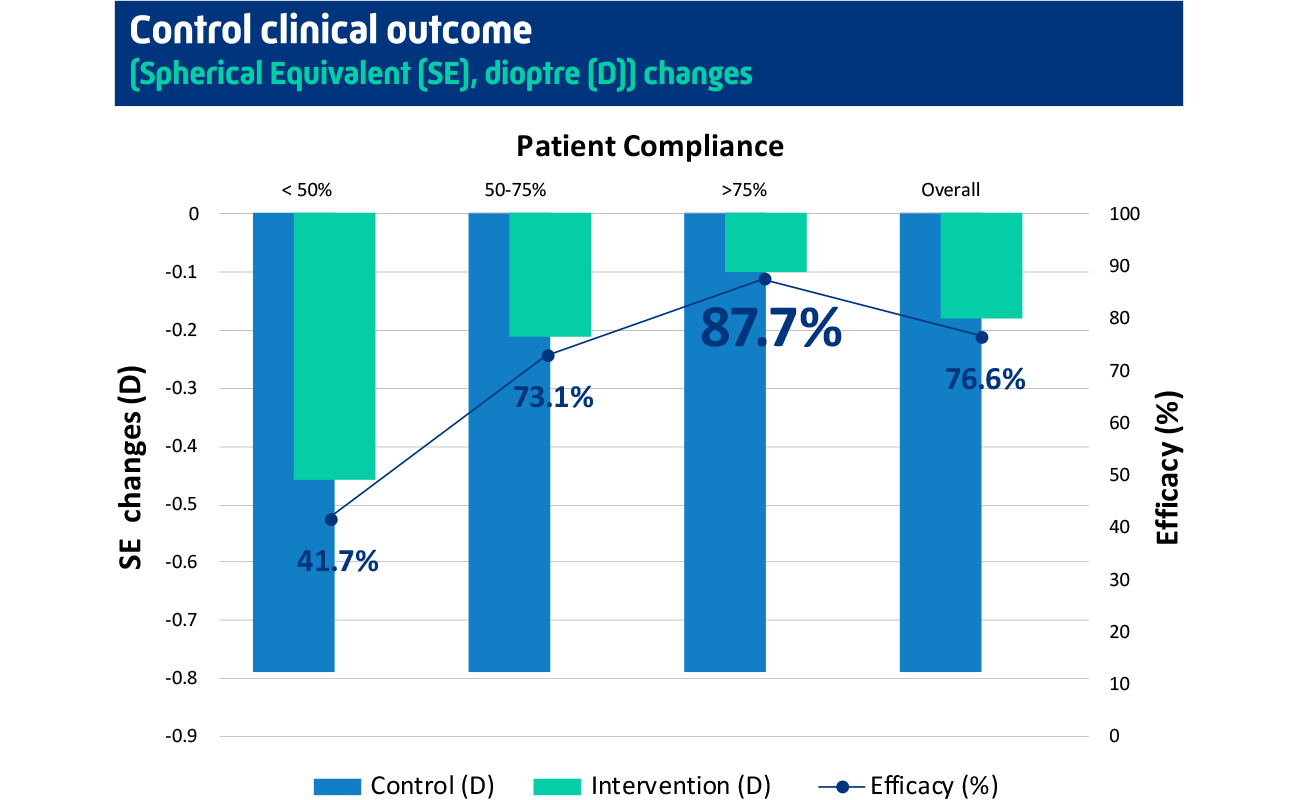
Achieving up to 87.7% myopia control efficacy7
The device has been proven in published clinical trials to be effective in the treatment of myopia, dramatically slowing its progression by up to 87.7%.
Frequently asked questions
References
- https://pubmed.ncbi.nlm.nih.gov/26875007/
- https://retinatoday.com/articles/2019-sept/myopia-a-global-epidemic
- Ruiz-Pomeda A, Pérez-Sánchez B, Valls I, et al. MiSight Assessment Study Spain (MASS). A 2-year randomized clinical trial. Graefes Arch Clin Exp Ophthalmol. 2018;256(5):1011-1021.
- Eyenovia enrolls first patient in phase III CHAPERONE Study for progressive myopia [news release]. New York: Globe Newswire; June 4, 2019. https://www.globenewswire.com/news-release/2019/06/04/1863931/0/en/Eyenovia-Enrolls-First-Patient-in-Phase-III-CHAPERONE-Study-for-Progressive-Myopia.html. Accessed June 25, 2019.
- Lam CSY, Tang WC, Tse DY, et al. Defocus incorporated multiple segments (DIMS) spectacle lenses slow myopia progression: a 2-year randomised clinical trial [published online ahead of print May 29, 2019]. Br J Ophthalmol. doi: 10.1136/bjophthalmol-2018-313739.
- Singapore National Eye Centre, Singapore Eye Research Institute, and Johnson & Johnson Vision set sights on halting global myopia epidemic [news release]. Singapore: PRNewswire; November 12, 2018. https://www.prnewswire.com/news-releases/singapore-national-eye-centre-singapore-eye-research-institute-and-johnson–johnson-vision-set-sights-on-halting-global-myopia-epidemic-300747895.html. Accessed June 25, 2019.
- Jiang, Yu, et al. “Effect of repeated low-level red-light therapy for myopia control in children: a multicenter randomized controlled trial.” Ophthalmology 129.5 (2022): 509-519.





